
Mounjaro
What is Mounjaro
Mounjaro (active ingredient: tirzepatide) is an injectable medication by Eli Lilly and Company. It was approved by Health Canada in November 2022 for improving glycaemic control in adults with type 2 diabetes mellitus.
The drug is the first-and-only in Canada that acts on both GIP (glucose-dependent insulinotropic polypeptide) and GLP-1 (glucagon‐like peptide-1) receptors. It is available as weekly injections via multi-dose pre-filled pen (KwikPen) or in some circumstances vials.[1]
Mechanistically, Mounjaro stimulates insulin release, suppresses glucagon, slows gastric emptying and reduces appetite-combined effects which help lower blood sugar and also lead to weight loss in many users. Some off-label or forthcoming uses include chronic weight management, but as of now the indication in Canada remains for diabetes.
A medicine with the same active ingredient is Zepbound. It was approved by Health Canada on May 13, 2025 for chronic weight management as an adjunct to a reduced-calorie diet and increased physical activity. The indication covers adults with BMI ≥ 30 kg/m², or BMI ≥ 27 kg/m² with at least one weight-related condition (e.g., hypertension, dyslipidaemia, prediabetes, type 2 diabetes, sleep apnoea, or cardiovascular disease).
Buying Mounjaro Online
In Canada, you can obtain Mounjaro via licensed telemedicine services and online pharmacies – but access still demands a valid prescription from a Canadian-licensed health-care provider. First you complete an online health assessment, then a remote consultation takes place evaluating your medical history, type 2 diabetes status, current medications and other relevant health data. If the provider deems it appropriate, a prescription is written and forwarded to a licensed Canadian pharmacy. The medication is then shipped to your home or local dispensing pharmacy. This digital method offers convenience and time savings for those in remote areas or with busy schedules. It’s not simply “click-and-get” without medical oversight – the online component is still a regulated medical process.
Mounjaro Without a Prescription?
Mounjaro is strictly a prescription-only medication in Canada. You cannot legally purchase it without a valid prescription from a licensed prescriber. While tele-health platforms streamline access, they do not equate to over-the-counter purchase. If a website claims to sell Mounjaro without any consultation or prescription, that likely falls outside Canadian regulations and may involve unsafe or counterfeit products. Legal access always involves assessment, prescription issuance and pharmacy dispensing under Canadian law.

Prices in Canada
Out-of-pocket cost for Mounjaro in Canada can be high. Insurance coverage is inconsistent: many public drug plans have not routinely covered Mounjaro and private-plan reimbursement depends heavily on plan specifics. It’s practical to verify cost and coverage before starting.
Another source lists vial pricing starting at CAD $546.99 – $906.99 for 1 pen injector (2.5 mg – 15 mg) in certain online listings – but true competitive retail price may vary.
Usage and Outcomes
Mounjaro is used once weekly by subcutaneous injection and in clinical trials for type 2 diabetes has shown reductions in HbA1c and body weight. Each pre-filled KwikPen is designed for single-patient use and delivers a fixed dose. Available strengths (per injection): 2.5 mg, 5 mg, 7.5 mg, 10 mg, 12.5 mg and 15 mg.
Start with 2.5 mg once weekly for the first 4 weeks. The maximum recommended dose is 15 mg once weekly. Each dose escalation depends on individual tolerance and blood-glucose response. Mounjaro should be injected subcutaneously in the abdomen, thigh, or upper arm, rotating sites each week.
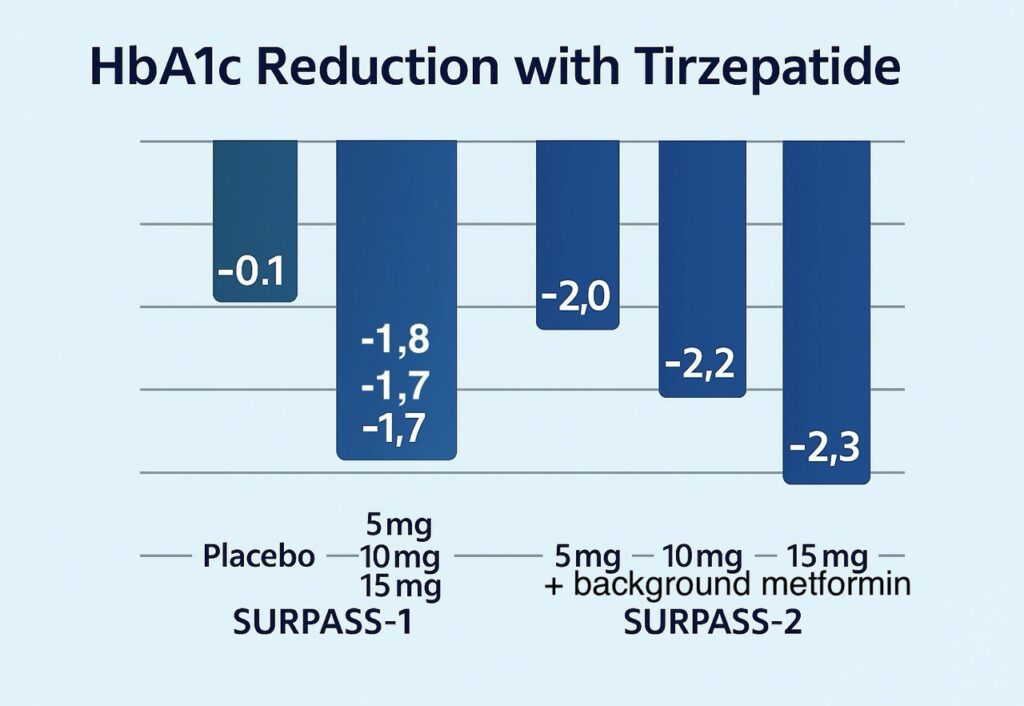
In SURPASS-1 (40 weeks, monotherapy), once-weekly tirzepatide at 5 mg, 10 mg, and 15 mg reduced HbA1c (glycosylated hemoglobin) by –1.8, –1.7, and –1.7 percentage points, compared with –0.1 percentage point for placebo.
In SURPASS-2 (40 weeks, with background metformin), tirzepatide 5 mg, 10 mg, and 15 mg achieved HbA1c reductions of –2.0, –2.2, and –2.3 percentage points, versus –1.9 percentage points.
Regarding weight outcomes: although not yet officially approved in Canada for obesity, the drug’s mechanism and trial data suggest meaningful weight loss occurs in many users. Real-world reports and comparative studies indicate outcomes may exceed older GLP-1-only medications. In practice, the real effect depends on consistent dosing, adherence, diet, exercise and monitoring. If someone expects rapid dramatic results without lifestyle change, they may be disappointed.
Side Effects and Contraindications
Mounjaro carries potential side-effects and contraindications you must know. Common adverse effects include gastrointestinal symptoms such as nausea, vomiting, constipation, diarrhoea and abdominal pain.
More serious risks exist: for example‐ potential for pancreatitis, gallbladder disease, and in specific populations risk of medullary thyroid carcinoma (MTC) if personal/family history, or multiple endocrine neoplasia type 2 (MEN2). It is contraindicated in those conditions. Additionally, patients with severe gastrointestinal disease, delayed gastric emptying or diabetic retinopathy (eye disease) should proceed with caution.
Dosing must be titrated gradually to mitigate side-effects. Because Mounjaro affects both GIP and GLP-1 receptors, its effects may be more potent – but also may pose greater risk of side-effects if not managed carefully.
Experience of People
Users report a mix of outcomes with Mounjaro. Some express that they felt less hunger, reduced cravings and steady drops in weight – comments:
- «I started Mounjaro and lost 12 pounds the first month… I’ve been on the medication for 6 months and have lost a total of 56 pounds. … This medication was life-changing for me.»[2]
- „Week 3 … No food cravings or hunger pangs.“[3]
- „In 5 months I am down over 60 pounds, and my insulin intake is down over 30%…”.[4]
Others say they were frustrated: side-effects slowed them down, or weight plateaued sooner than hoped and they stopped treatment. Real-world experience shows promise, yet highlights that medication alone may not “carry the load” without supporting changes.
Alternatives
If Mounjaro is unsuitable (cost, contraindications, access), consider alternatives. Other GLP-1 receptor agonists may be available. Older diabetes treatments (metformin, SGLT2 inhibitors) may not match the same effect but could be part of a broader plan. Non-drug alternatives remain fundamental: structured diet, exercise, behavioural therapy and surgical options may still play major roles. The best approach is aligned with your health profile, budget and long-term plan.
Mounjaro vs Ozempic
| Feature | Mounjaro | Ozempic |
|---|---|---|
| Active ingredient / class | Tirzepatide – dual GIP/GLP-1 receptor agonist. | Semaglutide – GLP-1 receptor agonist. |
| Health Canada status (primary indication) | Approved Nov 24, 2022 for type 2 diabetes (glycaemic control). | Approved for type 2 diabetes (glycaemic control); product monograph current to 2025. |
| Brand for weight management in Canada | Zepbound (tirzepatide) – separate brand approved for chronic weight management (May 13, 2025). | Wegovy (semaglutide) – separate brand approved for chronic weight management. |
| Dosing (diabetes) | Weekly 2.5 mg → 5 → 7.5 → 10 → 12.5 → 15 mg (step up – every 4 weeks). Pen strengths: 2.5–15 mg. | Weekly 0.25 mg (initiation) → 0.5 → 1.0 mg; some patients use 2.0 mg in practice per latest monograph. |
| Format | Prefilled KwikPen for subcutaneous injection. | Prefilled pen for subcutaneous injection. |
| Glycaemic and weight effects (head-to-head / trials) | In SURPASS-2, tirzepatide 15 mg lowered A1C more and produced – twice the weight loss vs semaglutide 1 mg. | SUSTAIN program: semaglutide reduced A1C and weight versus comparators; strong class efficacy.[5] |
FAQ
It is used for type 2 diabetes and, in some jurisdictions, for chronic weight management alongside diet and activity. Brand names and indications can differ by country (e.g., Zepbound for weight management)
Large trials show clinically meaningful weight reductions (often in the low-to-mid-teens percentage range over – 1.5 years), depending on dose and adherence.
It’s a subcutaneous injection once a week (abdomen, thigh, or upper arm). Doses are increased gradually to improve tolerability. Your prescriber sets the schedule.
There is a warning about thyroid C-cell tumours seen in animals. Do not use if you have a personal/family history of medullary thyroid cancer or MEN2. Pancreatitis and gallbladder issues have been reported – seek care for severe abdominal pain. Not for use in pregnancy.
It slows gastric emptying, which can affect absorption of oral medicines. Extra care is needed with drugs that have a narrow therapeutic index. For oral contraceptives, consider a non-oral method or add a barrier method for a period after starting or increasing the dose. Ask your clinician.
Yes, but doses of those medicines may need to be reduced to avoid hypoglycaemia. This requires medical supervision.
References
-
1. Eli Lilly and Company. PRODUCT MONOGRAPH INCLUDING PATIENT MEDICATION INFORMATION MOUNJARO (tirzepatide injection). Canada; [SEP 24, 2024]. Retrieved November 6, 2025
-
2. Mounjaro User Reviews & Ratings. Retrieved November 6, 2025
-
3. Reddit. 3 Weeks on Mounjaro: My Experience and Progress. Retrieved November 6, 2025
-
4. Diabetes a community of people touched by diabetes. Mounjaro. Retrieved November 6, 2025
-
5. Ahrén B, Atkin SL, Charpentier G, Warren ML, Wilding JPH, Birch S, Holst AG, Leiter LA. Semaglutide induces weight loss in subjects with type 2 diabetes regardless of baseline BMI or gastrointestinal adverse events in the SUSTAIN 1 to 5 trials. Diabetes Obes Metab. 2018 Sep;20(9):2210-2219. doi: 10.1111/dom.13353. Epub 2018 Jun 12. PMID: 29766634; PMCID: PMC6099440.






